for crossbridges to occur what events must happen
Chapter 19. The Musculoskeletal System
xix.4 Muscle Contraction and Locomotion
Learning Objectives
By the end of this department, you lot volition exist able to:
- Allocate the dissimilar types of muscle tissue
- Explain the role of muscles in locomotion
Musculus cells are specialized for contraction. Muscles allow for motions such as walking, and they also facilitate bodily processes such as respiration and digestion. The body contains three types of musculus tissue: skeletal musculus, cardiac muscle, and polish muscle (Effigy 19.33).
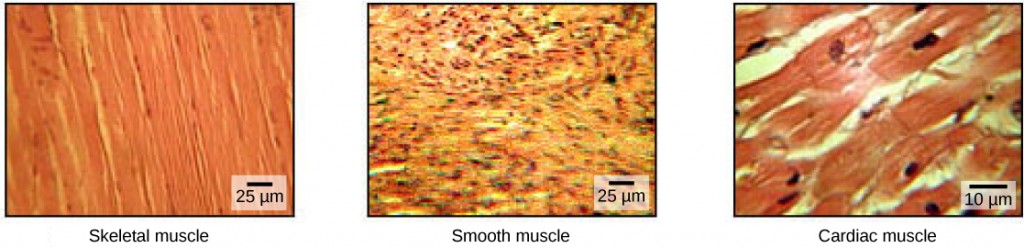
Skeletal musculus tissue forms skeletal muscles, which attach to bones or skin and control locomotion and whatever movement that tin can be consciously controlled. Considering it can be controlled by thought, skeletal muscle is likewise called voluntary muscle. Skeletal muscles are long and cylindrical in appearance; when viewed nether a microscope, skeletal muscle tissue has a striped or striated appearance. The striations are caused by the regular arrangement of contractile proteins (actin and myosin). Actin is a globular contractile poly peptide that interacts with myosin for muscle contraction. Skeletal muscle also has multiple nuclei nowadays in a unmarried jail cell.
Smooth muscle tissue occurs in the walls of hollow organs such every bit the intestines, breadbasket, and urinary bladder, and around passages such as the respiratory tract and claret vessels. Smooth muscle has no striations, is not nether voluntary control, has only one nucleus per prison cell, is tapered at both ends, and is called involuntary muscle.
Cardiac muscle tissue is only constitute in the centre, and cardiac contractions pump claret throughout the torso and maintain blood pressure. Like skeletal muscle, cardiac muscle is striated, but dissimilar skeletal muscle, cardiac muscle cannot exist consciously controlled and is called involuntary muscle. It has one nucleus per cell, is branched, and is distinguished by the presence of intercalated disks.
Skeletal Muscle Fiber Structure
Each skeletal muscle fiber is a skeletal muscle jail cell. These cells are incredibly large, with diameters of up to 100 µm and lengths of up to 30 cm. The plasma membrane of a skeletal musculus fiber is called the sarcolemma. The sarcolemma is the site of action potential conduction, which triggers muscle wrinkle. Within each musculus cobweb are myofibrils—long cylindrical structures that prevarication parallel to the muscle fiber. Myofibrils run the entire length of the muscle fiber, and because they are but approximately 1.2 µm in diameter, hundreds to thousands can be establish inside 1 muscle fiber. They attach to the sarcolemma at their ends, so that equally myofibrils shorten, the entire muscle cell contracts (Effigy 19.34).
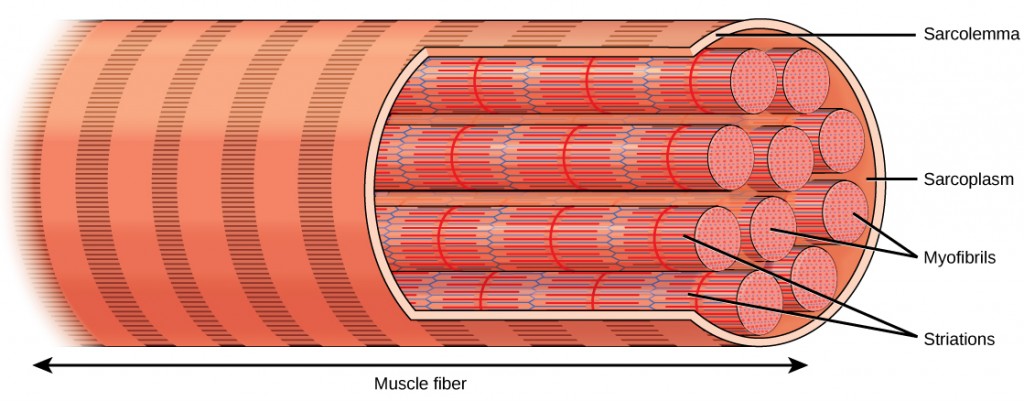
The striated appearance of skeletal muscle tissue is a result of repeating bands of the proteins actin and myosin that are present along the length of myofibrils. Dark A bands and lite I bands echo along myofibrils, and the alignment of myofibrils in the prison cell causes the entire cell to announced striated or banded.
Each I ring has a dense line running vertically through the middle called a Z disc or Z line. The Z discs mark the border of units called sarcomeres, which are the functional units of skeletal muscle. One sarcomere is the space between two sequent Z discs and contains one entire A band and two halves of an I band, one on either side of the A band. A myofibril is equanimous of many sarcomeres running along its length, and as the sarcomeres individually contract, the myofibrils and muscle cells shorten (Figure nineteen.35).
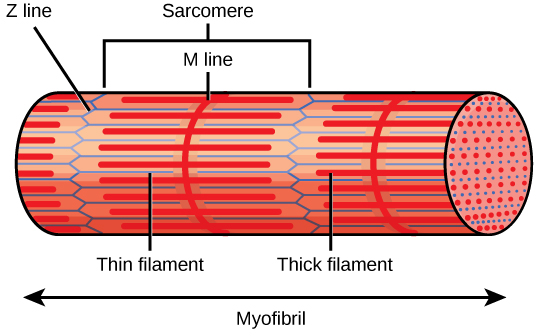
A sarcomere is the region from one Z line to the next Z line. Many sarcomeres are present in a myofibril, resulting in the striation pattern characteristic of skeletal muscle.
Myofibrils are equanimous of smaller structures called myofilaments. There are two main types of filaments: thick filaments and sparse filaments; each has different compositions and locations. Thick filaments occur only in the A ring of a myofibril. Thin filaments attach to a protein in the Z disc called alpha-actinin and occur across the unabridged length of the I band and partway into the A band. The region at which thick and thin filaments overlap has a dumbo appearance, as there is little space betwixt the filaments. Thin filaments do not extend all the way into the A bands, leaving a primal region of the A ring that only contains thick filaments. This central region of the A band looks slightly lighter than the rest of the A band and is chosen the H zone. The middle of the H zone has a vertical line called the M line, at which accessory proteins concur together thick filaments. Both the Z disc and the M line hold myofilaments in identify to maintain the structural arrangement and layering of the myofibril. Myofibrils are continued to each other by intermediate, or desmin, filaments that attach to the Z disc.
Thick and thin filaments are themselves composed of proteins. Thick filaments are composed of the protein myosin. The tail of a myosin molecule connects with other myosin molecules to course the central region of a thick filament near the M line, whereas the heads align on either side of the thick filament where the thin filaments overlap. The primary component of sparse filaments is the actin protein. Ii other components of the thin filament are tropomyosin and troponin. Actin has bounden sites for myosin attachment. Strands of tropomyosin block the binding sites and prevent actin–myosin interactions when the muscles are at rest. Troponin consists of three globular subunits. I subunit binds to tropomyosin, one subunit binds to actin, and ane subunit binds Ca2+ ions.
Concept in Action

View this animation showing the organization of muscle fibers.
Sliding Filament Model of Contraction
For a muscle cell to contract, the sarcomere must shorten. However, thick and thin filaments—the components of sarcomeres—do not shorten. Instead, they slide by one another, causing the sarcomere to shorten while the filaments remain the same length. The sliding filament theory of musculus wrinkle was developed to fit the differences observed in the named bands on the sarcomere at different degrees of muscle contraction and relaxation. The mechanism of contraction is the bounden of myosin to actin, forming cross-bridges that generate filament movement (Figure 19.36).
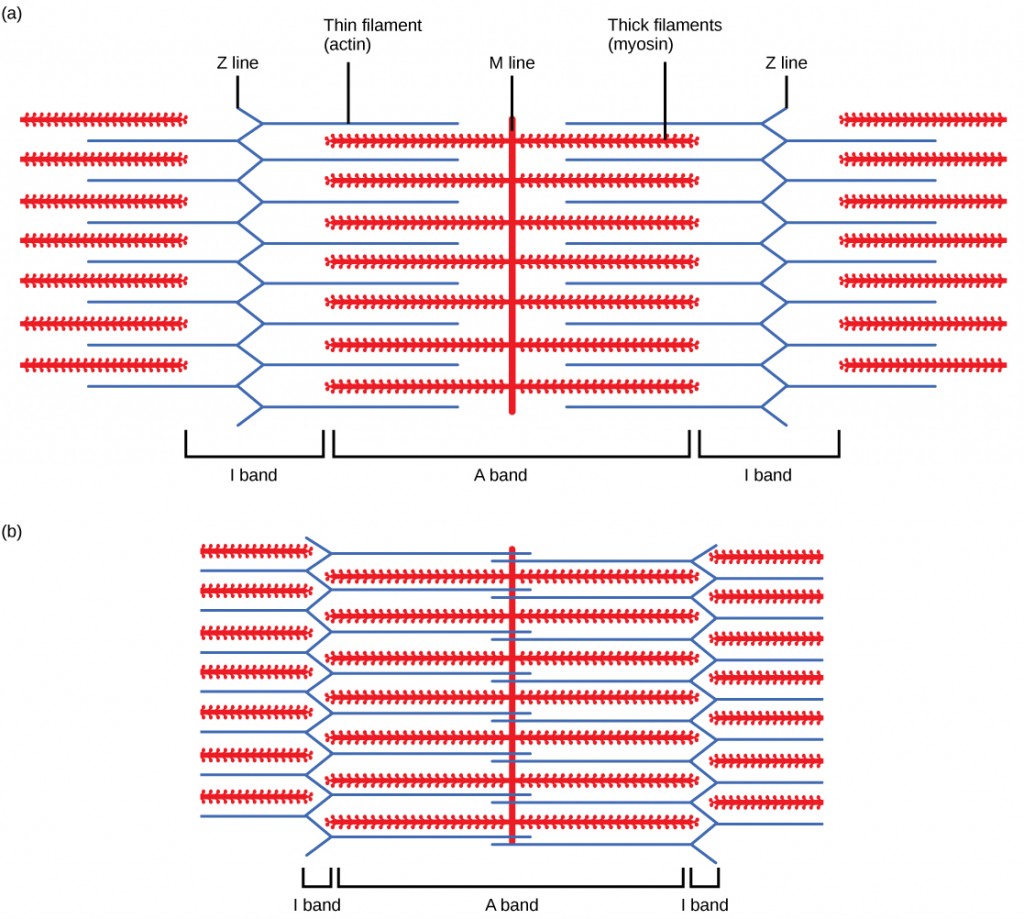
When (a) a sarcomere (b) contracts, the Z lines move closer together and the I band gets smaller. The A ring stays the same width and, at full contraction, the sparse filaments overlap.
When a sarcomere shortens, some regions shorten whereas others stay the same length. A sarcomere is defined every bit the distance between two consecutive Z discs or Z lines; when a muscle contracts, the distance betwixt the Z discs is reduced. The H zone—the key region of the A zone—contains only thick filaments and is shortened during contraction. The I band contains only thin filaments and besides shortens. The A ring does non shorten—it remains the same length—only A bands of different sarcomeres move closer together during wrinkle, eventually disappearing. Thin filaments are pulled by the thick filaments toward the middle of the sarcomere until the Z discs approach the thick filaments. The zone of overlap, in which thin filaments and thick filaments occupy the aforementioned expanse, increases as the sparse filaments motion inward.
ATP and Muscle Wrinkle
The motion of muscle shortening occurs every bit myosin heads bind to actin and pull the actin inwards. This activity requires energy, which is provided past ATP. Myosin binds to actin at a binding site on the globular actin protein. Myosin has another binding site for ATP at which enzymatic activity hydrolyzes ATP to ADP, releasing an inorganic phosphate molecule and energy.
ATP bounden causes myosin to release actin, allowing actin and myosin to detach from each other. Afterward this happens, the newly bound ATP is converted to ADP and inorganic phosphate, Pi. The enzyme at the binding site on myosin is called ATPase. The energy released during ATP hydrolysis changes the angle of the myosin head into a "cocked" position. The myosin head is then in a position for further move, possessing potential free energy, only ADP and Pi are still attached. If actin bounden sites are covered and unavailable, the myosin will remain in the high energy configuration with ATP hydrolyzed, only notwithstanding attached.
If the actin binding sites are uncovered, a cantankerous-bridge volition grade; that is, the myosin head spans the distance between the actin and myosin molecules. Pi is and then released, allowing myosin to expend the stored energy every bit a conformational alter. The myosin caput moves toward the M line, pulling the actin forth with it. As the actin is pulled, the filaments motility approximately 10 nm toward the M line. This movement is called the power stroke, as it is the stride at which strength is produced. As the actin is pulled toward the Grand line, the sarcomere shortens and the muscle contracts.
When the myosin head is "cocked," it contains energy and is in a loftier-free energy configuration. This free energy is expended as the myosin head moves through the power stroke; at the end of the ability stroke, the myosin caput is in a low-energy position. After the ability stroke, ADP is released; however, the cross-bridge formed is nonetheless in place, and actin and myosin are bound together. ATP can and then attach to myosin, which allows the cross-bridge cycle to commencement again and further musculus wrinkle can occur (Figure 19.37).
Concept in Action

Lookout this video explaining how a muscle contraction is signaled.
Which of the following statements about musculus contraction is true?
- The power stroke occurs when ATP is hydrolyzed to ADP and phosphate.
- The power stroke occurs when ADP and phosphate dissociate from the myosin head.
- The power stroke occurs when ADP and phosphate dissociate from the actin active site.
- The power stroke occurs when Ca2+ binds the calcium head.
Concept in Action

View this animation of the cross-bridge muscle wrinkle.
Regulatory Proteins
When a muscle is in a resting state, actin and myosin are separated. To go along actin from binding to the active site on myosin, regulatory proteins block the molecular bounden sites. Tropomyosin blocks myosin bounden sites on actin molecules, preventing cross-span germination and preventing contraction in a muscle without nervous input. Troponin binds to tropomyosin and helps to position information technology on the actin molecule; it also binds calcium ions.
To enable a muscle contraction, tropomyosin must modify conformation, uncovering the myosin-binding site on an actin molecule and allowing cantankerous-bridge formation. This can only happen in the presence of calcium, which is kept at extremely low concentrations in the sarcoplasm. If present, calcium ions demark to troponin, causing conformational changes in troponin that allow tropomyosin to movement away from the myosin binding sites on actin. One time the tropomyosin is removed, a cross-bridge can form between actin and myosin, triggering contraction. Cross-bridge cycling continues until Caii+ ions and ATP are no longer available and tropomyosin again covers the binding sites on actin.
Excitation–Contraction Coupling
Excitation–contraction coupling is the link (transduction) betwixt the activeness potential generated in the sarcolemma and the start of a muscle contraction. The trigger for calcium release from the sarcoplasmic reticulum into the sarcoplasm is a neural signal. Each skeletal musculus fiber is controlled by a motor neuron, which conducts signals from the brain or spinal cord to the muscle. The area of the sarcolemma on the muscle fiber that interacts with the neuron is chosen the motor terminate plate. The end of the neuron'south axon is chosen the synaptic terminal, and it does not actually contact the motor end plate. A pocket-sized infinite called the synaptic cleft separates the synaptic concluding from the motor end plate. Electrical signals travel along the neuron'southward axon, which branches through the muscle and connects to individual muscle fibers at a neuromuscular junction.
The power of cells to communicate electrically requires that the cells expend energy to create an electrical gradient across their jail cell membranes. This accuse gradient is carried by ions, which are differentially distributed beyond the membrane. Each ion exerts an electrical influence and a concentration influence. Merely every bit milk volition eventually mix with java without the need to stir, ions also distribute themselves evenly, if they are permitted to do so. In this case, they are not permitted to return to an evenly mixed country.
The sodium–potassium ATPase uses cellular free energy to motion 1000+ ions inside the cell and Na+ ions outside. This alone accumulates a small electrical charge, but a big concentration gradient. There is lots of K+ in the jail cell and lots of Na+ outside the jail cell. Potassium is able to leave the jail cell through One thousand+ channels that are open 90% of the time, and information technology does. Nevertheless, Na+ channels are rarely open, so Na+ remains outside the jail cell. When Thou+ leaves the cell, obeying its concentration gradient, that effectively leaves a negative charge behind. So at rest, there is a large concentration slope for Na+ to enter the prison cell, and there is an aggregating of negative charges left backside in the cell. This is the resting membrane potential. Potential in this context means a separation of electrical accuse that is capable of doing work. Information technology is measured in volts, only like a battery. However, the transmembrane potential is considerably smaller (0.07 V); therefore, the small value is expressed as millivolts (mV) or lxx mV. Because the inside of a prison cell is negative compared with the outside, a minus sign signifies the excess of negative charges inside the cell, −70 mV.
If an event changes the permeability of the membrane to Na+ ions, they will enter the cell. That will change the voltage. This is an electrical issue, called an activity potential, that can be used as a cellular signal. Communication occurs between nerves and muscles through neurotransmitters. Neuron activeness potentials cause the release of neurotransmitters from the synaptic terminal into the synaptic cleft, where they can then diffuse beyond the synaptic cleft and bind to a receptor molecule on the motor end plate. The motor end plate possesses junctional folds—folds in the sarcolemma that create a big area for the neurotransmitter to bind to receptors. The receptors are really sodium channels that open to allow the passage of Na+ into the cell when they receive neurotransmitter signal.
Acetylcholine (ACh) is a neurotransmitter released by motor neurons that binds to receptors in the motor end plate. Neurotransmitter release occurs when an action potential travels downward the motor neuron's axon, resulting in altered permeability of the synaptic terminal membrane and an influx of calcium. The Caii+ ions let synaptic vesicles to move to and bind with the presynaptic membrane (on the neuron), and release neurotransmitter from the vesicles into the synaptic crevice. Once released by the synaptic final, ACh diffuses beyond the synaptic crack to the motor end plate, where it binds with ACh receptors. As a neurotransmitter binds, these ion channels open, and Na+ ions cantankerous the membrane into the muscle cell. This reduces the voltage difference between the inside and outside of the cell, which is called depolarization. As ACh binds at the motor end plate, this depolarization is called an terminate-plate potential. The depolarization then spreads forth the sarcolemma, creating an activity potential equally sodium channels next to the initial depolarization site sense the change in voltage and open up. The activity potential moves beyond the entire cell, creating a wave of depolarization.
ACh is broken down past the enzyme acetylcholinesterase (Ache) into acetyl and choline. AChE resides in the synaptic cleft, breaking downwards ACh and then that it does non remain jump to ACh receptors, which would cause unwanted extended muscle contraction (Figure xix.38).
The deadly nervus gas Sarin irreversibly inhibits acetycholinesterase. What effect would Sarin have on muscle contraction?
Later depolarization, the membrane returns to its resting state. This is called repolarization, during which voltage-gated sodium channels close. Potassium channels continue at 90% conductance. Because the plasma membrane sodium–potassium ATPase always transports ions, the resting land (negatively charged inside relative to the outside) is restored. The period immediately post-obit the transmission of an impulse in a nerve or muscle, in which a neuron or muscle cell regains its ability to transmit another impulse, is chosen the refractory catamenia. During the refractory menstruation, the membrane cannot generate another action potential. . The refractory period allows the voltage-sensitive ion channels to render to their resting configurations. The sodium potassium ATPase continually moves Na+ dorsum out of the cell and K+ dorsum into the cell, and the K+ leaks out leaving negative charge behind. Very quickly, the membrane repolarizes, so that information technology can again be depolarized.
Control of Musculus Tension
Neural command initiates the formation of actin–myosin cross-bridges, leading to the sarcomere shortening involved in muscle contraction. These contractions extend from the muscle fiber through connective tissue to pull on bones, causing skeletal motility. The pull exerted by a muscle is called tension, and the amount of strength created past this tension tin can vary. This enables the same muscles to move very light objects and very heavy objects. In individual muscle fibers, the amount of tension produced depends on the cross-sectional area of the muscle fiber and the frequency of neural stimulation.
The number of cross-bridges formed between actin and myosin determine the corporeality of tension that a muscle fiber can produce. Cross-bridges can but form where thick and thin filaments overlap, assuasive myosin to demark to actin. If more cross-bridges are formed, more myosin volition pull on actin, and more than tension will be produced.
The ideal length of a sarcomere during product of maximal tension occurs when thick and thin filaments overlap to the greatest degree. If a sarcomere at rest is stretched past an ideal resting length, thick and thin filaments do non overlap to the greatest degree, and fewer cross-bridges can grade. This results in fewer myosin heads pulling on actin, and less tension is produced. As a sarcomere is shortened, the zone of overlap is reduced as the sparse filaments reach the H zone, which is composed of myosin tails. Considering information technology is myosin heads that class cross-bridges, actin volition non bind to myosin in this zone, reducing the tension produced by this myofiber. If the sarcomere is shortened fifty-fifty more, thin filaments begin to overlap with each other—reducing cantankerous-bridge formation even further, and producing fifty-fifty less tension. Conversely, if the sarcomere is stretched to the point at which thick and thin filaments practice not overlap at all, no cross-bridges are formed and no tension is produced. This amount of stretching does not ordinarily occur because accompaniment proteins, internal sensory fretfulness, and connective tissue oppose extreme stretching.
The primary variable determining strength product is the number of myofibers inside the muscle that receive an activity potential from the neuron that controls that fiber. When using the biceps to pick up a pencil, the motor cortex of the brain only signals a few neurons of the biceps, and only a few myofibers respond. In vertebrates, each myofiber responds fully if stimulated. When picking up a piano, the motor cortex signals all of the neurons in the biceps and every myofiber participates. This is shut to the maximum force the muscle can produce. As mentioned above, increasing the frequency of action potentials (the number of signals per second) can increase the force a bit more, considering the tropomyosin is flooded with calcium.
Summary
The body contains iii types of musculus tissue: skeletal musculus, cardiac muscle, and smooth musculus. Skeleton muscle tissue is equanimous of sarcomeres, the functional units of muscle tissue. Musculus contraction occurs when sarcomeres shorten, every bit thick and thin filaments slide past each other, which is called the sliding filament model of muscle wrinkle. ATP provides the energy for cross-span formation and filament sliding. Regulatory proteins, such as troponin and tropomyosin, control cross-bridge formation. Excitation–contraction coupling transduces the electrical signal of the neuron, via acetylcholine, to an electrical signal on the muscle membrane, which initiates force production. The number of muscle fibers contracting determines how much force the whole muscle produces.
Exercises
- Which of the post-obit statements nigh muscle contraction is true?
- The ability stroke occurs when ATP is hydrolyzed to ADP and phosphate.
- The ability stroke occurs when ADP and phosphate dissociate from the myosin caput.
- The power stroke occurs when ADP and phosphate dissociate from the actin active site.
- The power stroke occurs when Ca2+ binds the calcium head.
- The deadly nerve gas Sarin irreversibly inhibits acetycholinesterase. What result would Sarin take on muscle contraction?
- In relaxed muscle, the myosin-binding site on actin is blocked by ________.
- titin
- troponin
- myglobin
- tropomyosin
- The cell membrane of a muscle fiber is called a ________.
- myofibril
- sarcolemma
- sarcoplasm
- myofilament
- The muscle relaxes if no new nerve signal arrives. However the neurotransmitter from the previous stimulation is still present in the synapse. The activity of ________ helps to remove this neurotransmitter.
- myosin
- action potential
- tropomyosin
- acetylcholinesterase
- The ability of a musculus to generate tension immediately after stimulation is dependent on:
- myosin interaction with the M line
- overlap of myosin and actin
- actin attachments to the Z line
- none of the above
- How would muscle contractions be affected if ATP was completely depleted in a muscle fiber?
- What factors contribute to the amount of tension produced in an individual muscle fiber?
- What effect will low blood calcium have on neurons? What effect volition low claret calcium accept on skeletal muscles?
Answers
- B
- In the presence of Sarin, acetycholine is not removed from the synapse, resulting in continuous stimulation of the musculus plasma membrane. At get-go, muscle action is intense and uncontrolled, but the ion gradients dissipate, so electric signals in the T-tubules are no longer possible. The result is paralysis, leading to expiry by asphyxiation.
- D
- B
- D
- D
- Because ATP is required for myosin to release from actin, muscles would remain rigidly contracted until more ATP was available for the myosin cross-bridge release. This is why dead vertebrates undergo rigor mortis.
- The cantankerous-sectional surface area, the length of the musculus fiber at remainder, and the frequency of neural stimulation.
- Neurons will not exist able to release neurotransmitter without calcium. Skeletal muscles have calcium stored and don't need any from the outside.
Glossary
- acetylcholinesterase
- (Anguish) enzyme that breaks down ACh into acetyl and choline
- actin
- globular contractile protein that interacts with myosin for muscle contraction
- motor end plate
- sarcolemma of the muscle fiber that interacts with the neuron
- myofibril
- long cylindrical structures that lie parallel to the muscle fiber
- myofilament
- small structures that brand up myofibrils
- myosin
- contractile protein that interacts with actin for musculus contraction
- osseous tissue
- connective tissue that constitutes the endoskeleton
- sarcolemma
- plasma membrane of a skeletal muscle fiber
- sarcomere
- functional unit of measurement of skeletal muscle
- skeletal musculus tissue
- forms skeletal muscles, which attach to bones and control locomotion and any movement that can be consciously controlled
- spongy bone tissue
- forms the inner layer of all bones
- thick filament
- a group of myosin molecules
- thin filament
- 2 polymers of actin wound together along with tropomyosin and troponin
- tropomyosin
- acts to block myosin binding sites on actin molecules, preventing cantankerous-span formation and preventing contraction until a muscle receives a neuron bespeak
- troponin
- binds to tropomyosin and helps to position it on the actin molecule, and also binds calcium ions
- ulna
- bone located on the medial aspect (pinky-finger side) of the forearm
- vertebral column
- (also, spine) surrounds and protects the spinal cord, supports the head, and acts as an zipper signal for ribs and muscles of the back and neck
Source: https://opentextbc.ca/biology/chapter/19-4-muscle-contraction-and-locomotion/
0 Response to "for crossbridges to occur what events must happen"
Post a Comment